Ani Grigoryan
Molecular Medicine

Hartmut Geiger
Molecular Medicine
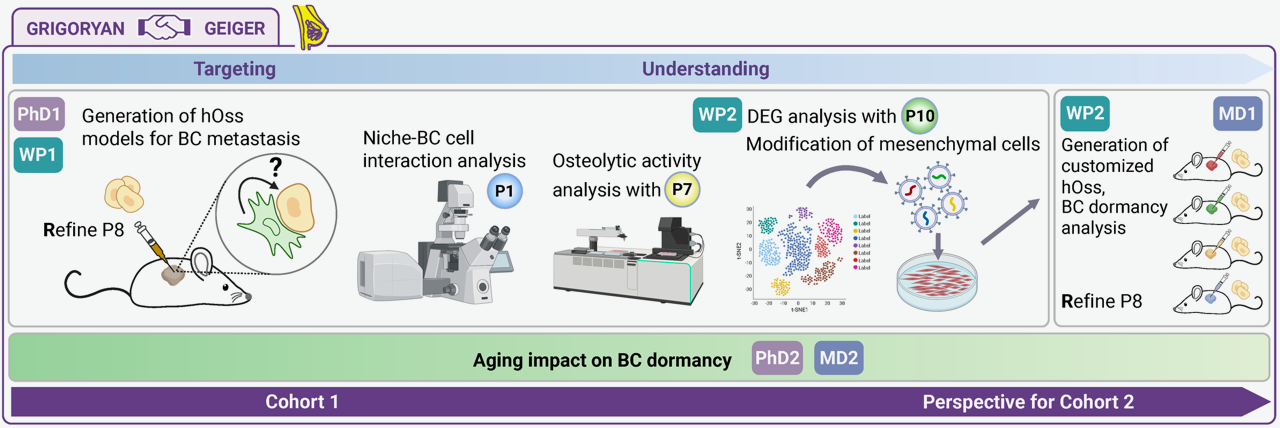
This project establishes patient-derived breast cancer organoids within humanized bone marrow niche models using decellularized ex vivo scaffolds (e.g., humanized ossicles or porcine bone matrices) to elucidate mechanisms of metastatic dormancy and reactivation in the bone microenvironment. Doctoral researchers will derive PDO lines from primary tumors and bone metastases of ER+/HER2- and triple-negative breast cancer patients, co-culture them with bone marrow mesenchymal stromal cells (BMSCs), osteoblasts, osteoclasts, and hematopoietic cells to recapitulate the dormancy niche, and perform longitudinal imaging and perturbation experiments tracking dormant cancer cell survival (Ki67low/CDK4low), symmetric/asymmetric division, and reactivation triggers (e.g., endocrine therapy withdrawal, bone turnover signals). Single-cell multiomics, live-cell imaging (light-sheet microscopy), secretome analysis, and CRISPR screens will map dormancy-inducing pathways (e.g., NR2F1/THY1 quiescence, BMP7/ TGFβ), immune surveillance evasion, and niche-derived soluble factors/cytokines promoting metastatic outgrowth. Patient-correlated validation identifies biomarkers predicting dormancy duration and tests pharmacological interventions (e.g., dormancy disruptors, niche reprogramming) to prevent bone relapse and improve metastatic breast cancer outcomes.